Isothiazolinone Biocides
Part 2 of our series, A Biocide Analysis, examines CMIT/MIT chemistry using the same 6 attributes: solubility, potency, compatibility, acceptability, stability and handleability. Since its introduction in the late 1970’s as a preservative and popularization in the 1980’s, CMIT/MIT has been used in varied applications including water, cosmetics and fuel treatment. As a preservative, the product has been touted by the manufacturer as the most widely used and most effective. While its wide range of use is acknowledged, this assessment will look at its effectiveness and application as a fuel biocide. Originally developed by Rohm and Haas, it is now produced by a few manufacturers and repackaged under numerous brand names.
Most Common CMIT/MIT Fuel Biocide Brands
- Kathon FP 1.5 (original brand no longer available – pulled from market by manufacturer)
- Kathon Fuel 15 Biocide (renamed FP 1.5 product after a 787 Boeing aircraft incident)
- FQS 1.5 Microbicide (7.3% less active ingredient than original Kathon product)
- Innospec Predator 8000
- Fueltreat FT-400
- Fueltreat BC-250
The product contains a mixture of the active ingredients (CMIT) 5-chloro-2-methyl-2H-isothiazol-3-one and (MIT) 2-methyl-2H-isothiazol-3-one in a 3 to 1 ratio, often referred to as CMIT/MIT. It is a member of a group of chemicals known as isothiazolinone. The fuel biocide mixture includes CMIT/MIT(1.5%) as well as magnesium dinitrate (salt), magnesium chloride (salt), water and dipropylene glycol (90%).
SOLUBILITY
Solubility is one of the most important characteristics of a biocide. If a biocide cannot properly mix and remain in the fuel, it will not be able to provide adequate decontamination nor protect a fuel system during long-term storage. Solubility issues can contribute to serious problems, like engine failure and microbial adaptation leading to catastrophic events. One such event happened when a CMIT/MIT product known as Kathon FP 1.5 was added to the fuel on a Boeing 787 aircraft with 301 passengers on board. The engines lost thrust forcing the aircraft to make an emergency landing. What caused the dangerous incidence? The insolubility of the CMIT/MIT biocide. As a result of this and other solubility related incidences, the product was banned from aviation use by aircraft manufacturers as well as global aviation authorities. The manufacturer at the time removed the product from the market.
Solubility issues happen more often than anyone realizes. Like many fuel biocides, Kathon was designed for water applications, not for fuel. It is miscible in water (100% water soluble) but offers very poor fuel solubility. The graph below illustrates the difference in solubility between CMIT/MIT and Biobor JF.
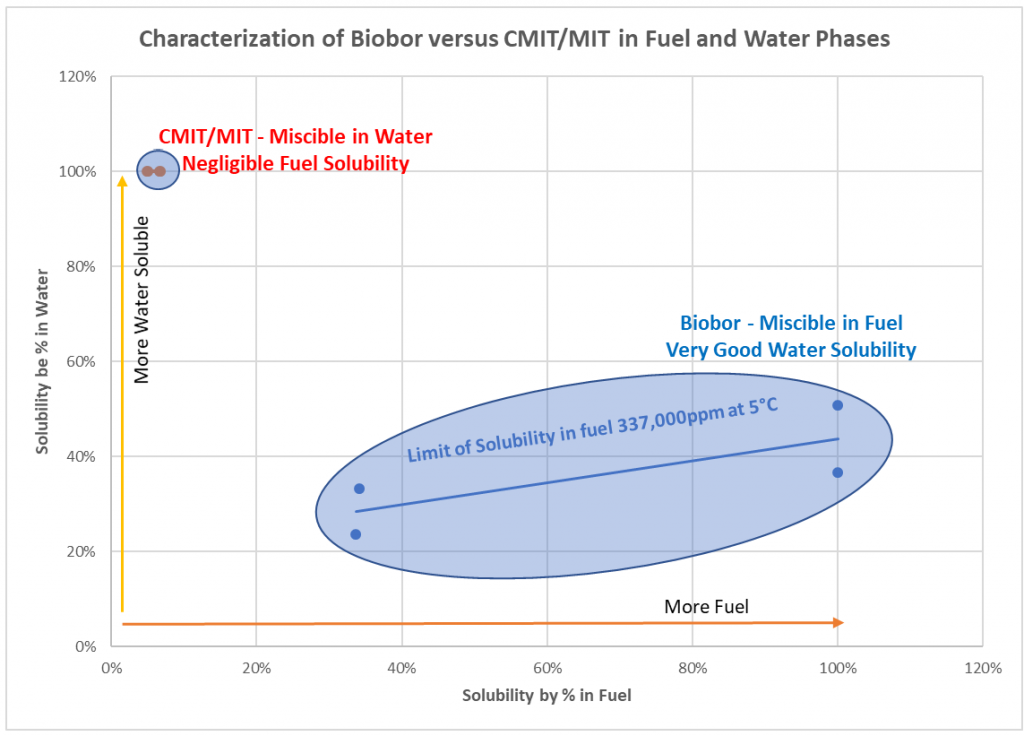
You will notice from the graph that Biobor JF is miscible in fuel and very soluble in water, making it the ideal fuel biocide. A recent laboratory study showed CMIT/MIT exhibits only around 5% fuel solubility, negligible at best. The graph above illustrates the difference between the two biocides.
“The active ingredient in Kathon FP 1.5 microbicide is completely soluble in water. Note that the product solvent, dipropylene glycol, is not completely soluble in fuel.”
The Manufacturer’s literature refers to kathon’s fuel solubility issues
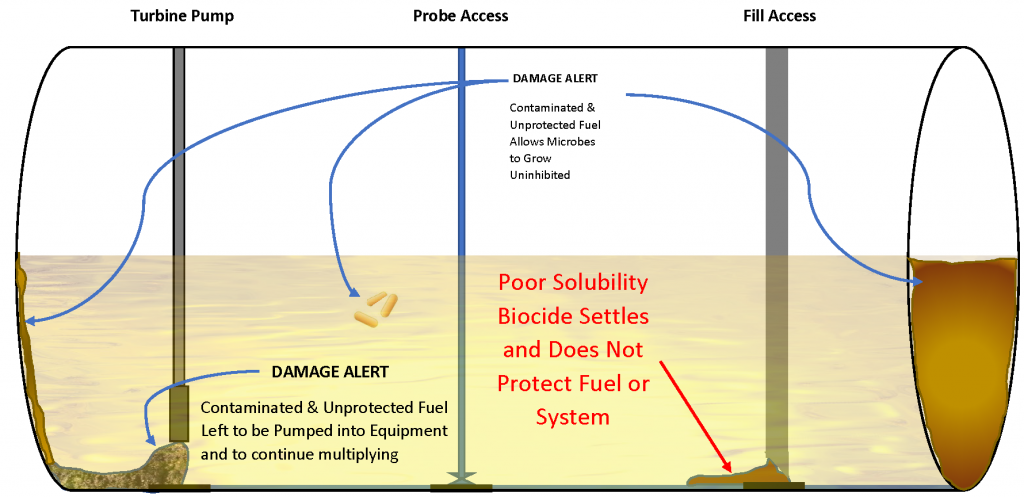
The contrast between Biobor JF fuel biocide and CMIT/MIT “water biocide” is plain to see. Kathon is fully water soluble, leaving a fuel system unprotected and Biobor JF is 100% soluble, protecting 100% of the fuel and the system it touches. In more complex systems, like generator tanks or baffled supplier tanks, the insoluble biocide has little chance of performing well, leaving the system unprotected against microbial contamination. The clear winner is Biobor JF.
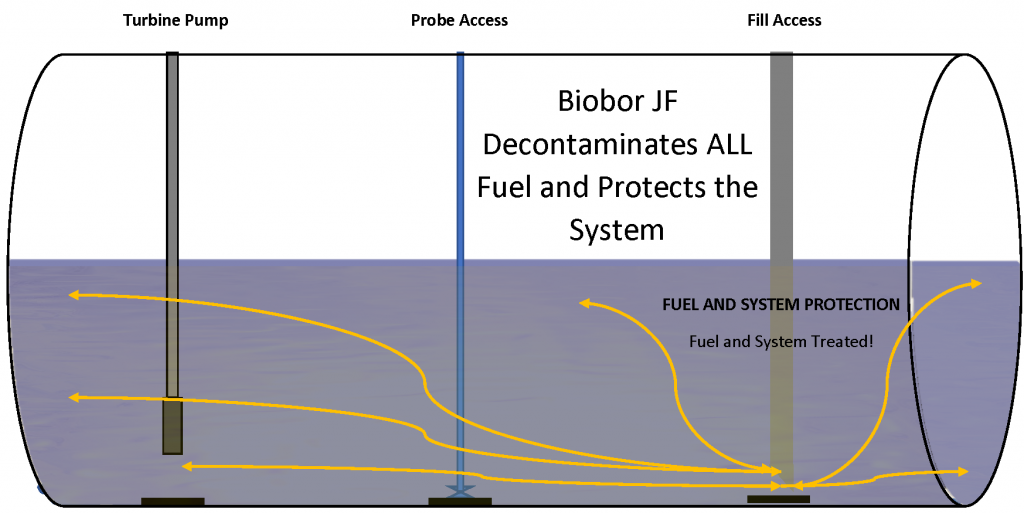
With any additive that exhibits solubility issues, product mixing is of paramount importance, likely the reason product instructions specifically relate effectiveness with mixing. Product application information notes “that effective treatment will only be achieved with efficient mixing.” A biocide that has a solubility gap, like CMIT/MIT, will have an efficacy issue regardless the dose rate. Solubility gaps and inadequate mixing result in unprotected fuel. If a product is prone to settle out of solution, the only area being protected is the small area where the product has settled. The rest of the fuel and system are left without adequate treatment where microorganisms will multiply and flourish. It is also possible to have inaccurate test results if the biocide has settled to a low point of access where samples are regularly taken. That location would have a higher concentration of biocide than the rest of the tank (potentially a majority of the biocide), showing a more efficacious result, but only for that very small area in the fuel system. The rest of the fuel and system are left unprotected.
POTENCY
Potency includes a review of the products efficacy (ability to kill an organism), adaptation to microbes, the potential for CMIT/MIT to be used as a microbial nutrient source and its ability to control biomass. To understand the actual efficacy of CMIT/MIT, recommended dose rates and soak times need to be examined. Manufacturer literature claims the product eradicates microbial growth in contaminated fuel systems in 24 to 72 hours. The efficacy of the product will be challenged based on their claim. According to the manufacturers product information, there are three dose rates the claim are effective.
CMIT/MIT Manufacturer Dose Rates
- 100-150ppmv – preventative/maintenance dose – for fuels that lack microbial contamination. The literature notes to exercise “extreme care” “to avoid the addition of a preventative/maintenance level dosage” to heavily contaminated fuel systems. Other manufacturers literature notes that this dosage should never be used if microbial contamination is present at any level.
- 200-300ppmv – curative dose – with the evidence of microbial contamination.
- 400-1000ppmv – shock dose – for fuels with heavy microbial contamination (2-5 times the recommended Biobor JF shock dose)
While the original manufacturer of CMIT/MIT claims the biocide is effective at lower dose rates, their own data shows otherwise. The chart taken from an original manufacturer study indicates the minimal biostatic dose rate for individual microbial organisms tested. Biostatic is not the same as biocidal. Biostatic relates to inhibiting growth but not killing the microbe. It can still multiply, just at a reduced rate. The concern for any sub-lethal, biostatic dosing of a biocide is microbial adaptation, resistance and microbial recontamination. There are well documented cases of CMIT/MIT adaptation and resistance. Research shows the regular application of sub-lethal doses of this biocide leads to microorganism resistance, rendering the product ineffective. Documented research validates the concern. Looking at the biostatic chart, it is easy to see that the manufacturer prescribed curative dose will not be effective against a host of known microbial contaminants. According to manufacturer documentation a large shock dose of 1000 ppm will be required, making it a considerably costlier treatment (5 times more than Biobor JF).

Inadequate mixing also affects soak times. The published soak time for CMIT/MIT is 24 to 72 hours depending on the level of microbial contamination. The product information specifically states that mixing methods “extend the treatment time required.” Documented efficacy data reveals the necessity for extended soak times and higher dose rates under all conditions tested
Additional studies reveal poor sub-lethal results. The ongoing concern when using CMIT/MIT in sub-lethal doses has to be adaptation and resistance. Even with increased soak times, microbial infestation still remains at high enough levels to create concern and cause problems. This does not bode well for fuel protection in long-term storage. That level of microbial contamination left in the fuel tanks will likely increase both maintenance cost and risk. Are higher doses of CMIT/MIT more effective? Does the 400-1,000ppmv shock dose eradicate microbes in the fuel? The manufacturer states that “a shock dose is required to decontaminate an already contaminated system.” This should be enough to dissuade the use of anything less when microbes are present. However, the question still remains, “Will a shock dose of CMIT/MIT eradicate microbial contamination?” In a 60-day efficacy study, it was found the biocide had only “a transitory inhibitory effect.” At 400ppmv it took over 7 days for the biocide to begin working on various biodiesels with low contamination levels containing natural water bottoms. Results showed only an inhibitory effect in all fuel types. The study used the reduction of biomass production to determine the biocides effectiveness. The table below shows the results by product type for both the low and high challenge.
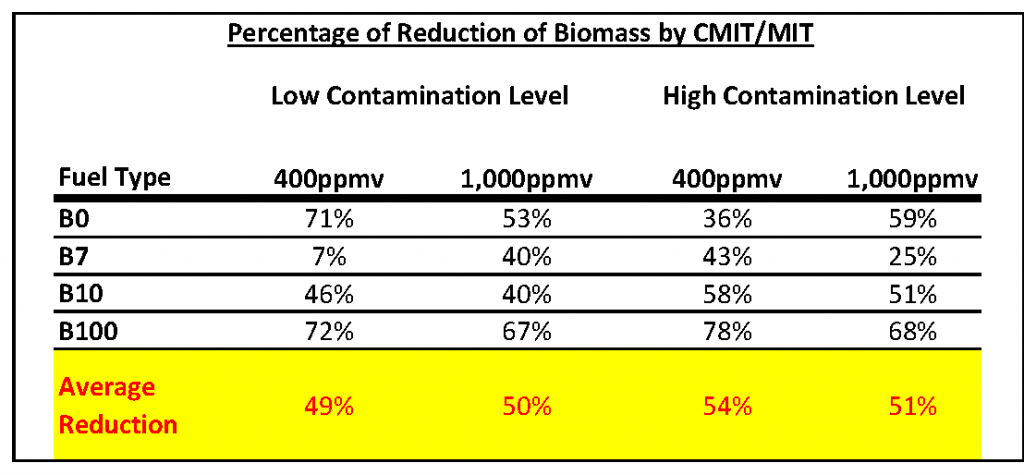
While there was a reduction in biomass, neither the 400ppmv or 1,000ppmv dose rates were able to eradicate the microbial contamination as illustrated by the continued biomass production. The aggregate reduction percentage was only 51% indicating a biostatic effect. Neither the 400ppmv or 1,000ppmv dose rates were capable of eradication as indicated by the continued biomass production. The researchers noted the “poor performance” of the biocide.
The manufacturer’s claim that CMIT/MIT eradicates microbial growth is overstated at best and unfounded in both its own data and third-party studies. According to the data, serious consideration should be given prior to any sub-lethal use of any CMIT/MIT brand. The biostatic graph data reinforces the limitation of the product.
COMPATIBILITY
How compatible is CMIT/MIT with fuel and fuel system components. The most glaring incompatibility issue stems from its lack of solubility in fuel. It is not compatible with fuel based on its solubility problem. Wherever it precipitates to is a potential problem. According to the manufacturer’s information, the active ingredients have a pH of 1.9.

That’s more acidic than sulfuric acid. That is acidic enough to eat through most materials. So how compatible is CMIT/MIT with fueling component materials? The chart shows some of the main materials likely to be found in fuel systems that CMIT/MIT is not compatible with. Biobor JF is not a corrosive material with broad incompatibilities like CMIT/MIT.
ACCEPTABILITY
While CMIT/MIT has wide acceptability as a water biocide, its acceptability as a fuel biocide has come into question since its serious problems in the aviation industry. The solubility problem raises so many questions and has a ripple effect into other industries. Fact, the formerly approved aviation fuel biocide, Kathon FP 1.5 (the original CMIT/MIT brand), was banned from aviation use in numerous aircraft. The manufacturer further decided to remove it from the market. Over more than 24 months aviation organizations, OEMs and government agencies around the world have reviewed and updated its publications due to Kathon’s problems. CMIT/MIT no longer has wide acceptability as a result. In contrast, Biobor JF is left as the only approved and available aviation fuel biocide. It may only be a matter of time before other industries do the same.
Another issue relates to the products sulfur content. Testing verified it contains around 3,500 ppm of sulfur potentially causing the fuel to be out of spec. Increasing the sulfur content in your fuel results in higher equipment maintenance. Studies show that long term use of a high sulfur product will increase the cost of vehicle operation by as much as 2 cents per gallon. Over the life of an engine, the additional cost can amount to thousands of dollars. One of the most significant reasons to abandon using high sulfur additives is the corrosive wear caused by sulfur in fuel. Corrosion is a serious problem amounting to shorter engine life. Sulfur increases corrosion wear and contributes to increased deposits. A separate study of Kathon in ULSD showed it actually reduced the lubricity of the fuel. Biobor JF does not contain sulfur and in contrast to CMIT/MIT is a lubricity additive.
STABILITY
The stability of a fuel biocide is important. The two most prevalent concerns affecting CMIT/MIT are pH and temperature. Both are shown to influence accelerated degradation, deactivation and violent thermal decomposition of the active ingredients.
Most bottom water environments have a pH range between 6.8 and 8.5. CMIT/MIT’s poor fuel solubility and high water solubility tends to drive the product to any bottom-water. In the case of fuel tanks, this is where CMIT/MIT biocide settles indiscriminately. Research shows that CMIT/MIT does not do well in a pH greater than 7. A pH of 7 is neutral, lower pH is acidic and higher pH is alkaline. The effectiveness of a biocide is determined by its ability to control microbial contamination. Any degradation over time reduces its effectiveness. There have been several studies of CMIT/MIT addressing the issue of pH. Some have recorded deactivation at a pH as low as 7. A detailed study showing the effect of pH on the degradation of CMIT/MIT notes “the active component undergoes degradation in alkaline solutions, and the rate of degradation is faster with an increase in pH.” It further suggests that the degradation of the biocide “results from hydrolysis of the chlorinated isothiazolone” (CMIT) as a result of the reaction to an alkaline pH. Alkaline pH continued to reduce the effectiveness of this biocide until complete deactivation. For long-term storage this presents a major problem. Over a short period of time, CMIT/MIT would deactivate and no longer provide protection to the fuel system.
Temperature also plays a significant role in the degradation and deactivation of CMIT/MIT. The manufacturer recommends avoiding temperatures above 40˚C (104˚F). It goes on to warn “the active ingredients can decompose violently at temperatures above 50˚C (122˚F). Generation of gases (hydrogen chloride, nitrogen oxides, sulfur oxides) can cause rapid pressure buildup in closed systems,” such as closed bottles or fuel systems. The dangers are obvious in the manufacturer’s warning. One study found that the active ingredients became unstable and began to deactivate at temperatures as low as 24˚C (75˚F). As the temperature goes up, the degradation and deactivation of the active ingredients accelerates.
The graph illustrates the deactivation of CMIT/MIT based on temperature and thermal decomposition. According to the graph, the CMIT/MIT would be completely deactivated at 60˚C (140°F) in four days. At 40˚C (104°F) deactivation would take just over 25 days and at 20˚C (75˚F) the deactivation would be 100% in one-year, possibly the reason the manufacturer has a published one-year shelf life on the product.
This data is significant especially for the protection of fuel in above ground storage tanks or the belly tank of a generator system. he correlation between high temperature exposure in these situations and the deactivation of the product is important to understanding.
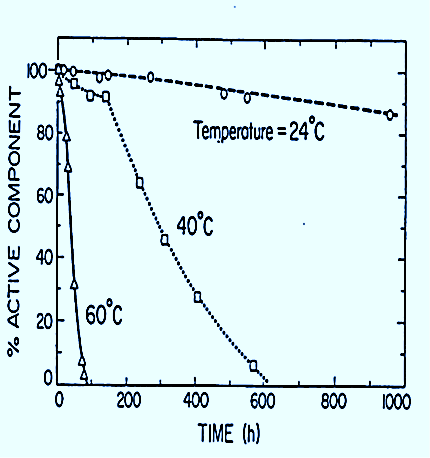
The reduced effectiveness of the biocide and the storage limitations as it pertains to higher temperatures play a role in whether or not it works effectively or potentially not at all. The higher the temperature, the shorter the shelf life of the product due to thermal decomposition. The chart below shows how outside air temperatures relate to inside storage temperatures.
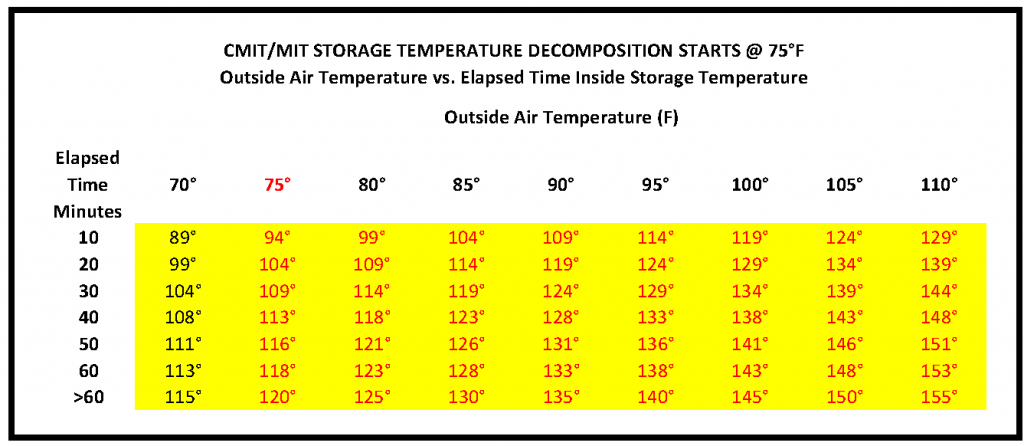
CMIT/MIT thermal decomposition begins at 75°F. Higher temperatures accelerate deactivation, reducing its efficacy, eventually rendering it completely ineffective. In contrast, Biobor JF is both pH and temperature stable.
HANDLEABILITY
Handleability relates directly to a biocide’s toxicity level. How toxic is too toxic? CMIT/MIT is under global scrutiny for its toxicity and serious implications to human health and the environment. Product documentation warns of its danger including chemical burns, allergic reactions and respiratory and lung problems due to exposure. There are hundreds of studies and peer reviewed journal articles documenting the serious health concerns attributed to exposure and use of the chemical. Over the last few years, there have been growing concerns among medical professionals and researchers due to the rapid increase of serious health issues associated with CMIT/MIT.
The biocide is identified as a contact allergen responsible for contact dermatitis. Between 2010 and 2012 nationwide testing in France was performed on over 7874 patients. The study showed a significant increase in positive test results for contact dermatitis indicating a rapidly emerging allergen.
A 2017 study confirmed the significant damage to the airways due to exposure to CMIT/MIT inhalation. Twenty-four patients were identified as having been exposed to the chemical. None had underlying diseases. Inhalation exposure of CMIT/MIT is just now coming to the forefront. There have been over 500 additional studies exposing inhalation danger. It was determined that inhalation exposure to the chemical was a direct cause of fatal lung injury. The results were reported in Korea from 2006 to 2011. Since then numerous other reports have been made on the deleterious effects of CMIT/MIT on humans.
The occupational use of CMIT/MIT has been tied to neurotoxicity. The chemical is highly toxic to human neurons (the basic working unit of the brain, the spinal cord, the central neural tracts and the peripheral nerves). Both acute and chronic consequences have been reported for humans exposed to the toxins. In the study, the chemical induced cell death as well as excessive DNA damage.
There is no doubt that the evidence is mounting on the toxicity of CMIT/MIT. Other effects of exposure to the chemical include hair loss – a sign of endocrine hormone disruption, inflammatory disease, and neuromuscular paralysis. Physical contact with CMIT/MIT, including inhalation puts the user at risk. The manufacturer warns, “Wear goggles or face shield and rubber gloves when handling. Avoid breathing vapor or mist.” The warning leaves no doubt how difficult handling this biocide can be.
The assessment of CMIT/MIT reveals a product that is long on promises and short on delivery. The manufacturer’s claim that the product eradicates microbial contamination is troubling. The results show limited biostatic affect in recommended dose rates. Even at a shock dose, the efficacy was not as expected from the claims made. Insolubility makes it difficult for the biocide to reach all areas of the fuel system to make an effective kill. Product stability is another concern. Both pH and temperature have a significant degradational effect. Dramatic increases in degradation are compounded when both pH and temperature work together to deactivate the biocide. Finally, toxicity has to play a role in determining the risk associated with its handling and use. Exposure to a chemical so toxic increases the likelihood for health related incidents. When a less toxic, proven biocide is available, why use this one? Use Biobor JF!
Follow BioborShare the Post








